There have been many theories on why adhesives can produce bonds, and there is no definitive answer to this point in the academic community until today. From a practical point of view, there is still no single adhesive theory that can explain all bonding phenomena. For hot melt adhesive films, what theory can explain the bonding? Let's see what theories there are for bonding.
The mechanical theory is also called the glue nail theory. In layman's terms, the glue is cured by penetrating the voids on the material's surface, forming a small glue nail, resulting in a mechanical bond, which makes the adhesive cured and the substrate mechanically fixed together. Because the solid does not behave perfectly smooth, the surface of the solid under the microscope consists of numerous irregular peaks and valleys.
The mechanical occlusion of the adhesive is the main bonding factor for multi-hollow base materials (e.g., foam). For the bonding of non-porous materials, mechanical sanding is much more effective than untreated. We often use this method of mechanical sanding in leather bonding and rubber bonding.
Mechanical roughening, i.e., sanding, has the following advantages.
1)formation of mechanical locking;
2) formation of a new surface;
3) formation of a highly reactive surface;
4) increase of the surface area.
The mechanical theory explains the bonding mechanism of rough surfaces well, but it is difficult to explain the bonding mechanism of smooth surfaces, which is its shortcoming.
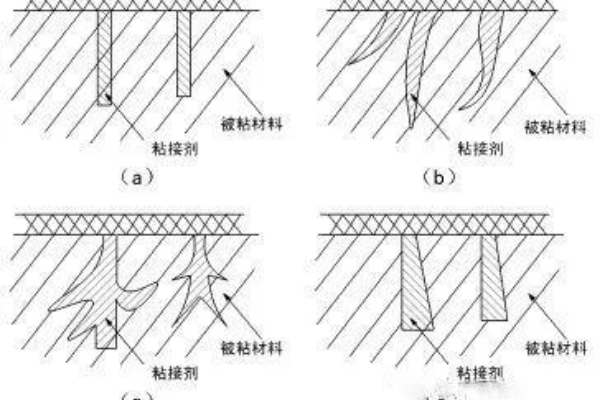
Adsorption theory believes that bonding is caused by molecular contact between two materials and produces surface forces. The process of contact between the adhesive and the surface of the bonded material is called "wetting." For the adhesive, the surface tension should be lower than the critical surface tension of the solid to achieve the effect of wetting.
Most organic adhesives are easy to wet metal solids. But many solid organic substrate surface tension is less than the common adhesive. Good wetting requires adhesives to have a lower surface tension than the substrate. Part of the organic adhesives (epoxy adhesives) have good adhesion to metals but poor adhesion to untreated polymer substrates, such as polyethylene, polypropylene, and fluorocarbons.
Adhesives on the sticky material wetting contact, through the intermolecular interaction forces can achieve permanent bonding, in the bonding cohesion can generate four types of chemical bonds.
Primary valence Jian has an ionic bond, covalent bond, metal parts. Secondary bonds are van der Waals forces.
The electrostatic theory believes that bonding exists because of a double electric layer in the interface between the adhesive and the bonded material, resulting in the formation of electrostatic forces. This electrostatic force itself is resistant to separation. People through the substrate have been peeled off the cured adhesive discharge phenomenon, confirming the validity of this theory.
Diffusion theory that the existence of adhesive force because of the mutual diffusion between the adhesive and bonded molecules and produced. When the adhesive and the bonded material are polymers, this theory is justified in the case of long molecular chains with movement. For example, solvent-based bonding or hot melt bonding of thermoplastics is the result of intermolecular diffusion.
Although the question of the bonding mechanism has never been settled, understanding the various bonding theories helps us grasp the basic bonding requirements. Perhaps bonding is not explained by one theory in the first place. Different bonding theories should be used for different materials for hot melt films, e.g., different theoretical models will apply for bonding on textiles and plastics. Among the various bonding mechanisms, the mechanical and diffusion theories are closer to the actual situation of hot melt film bonding.
Related articles:
What Types of Hot Melt Adhesive Films Are Commonly Used?
How do I use hot melt adhesive film?